NaI
NaI(Tl) or sodium iodide doped with thallium: NaI(Tl) is by far the most widely used scintillator material. It is available in single crystal form or the more rugged polycrystalline form (used in high vibration environments, e.g. wireline logging in the oil industry). Crystals with a higher level of doping are used in X-ray detectors with high spectrometric quality. Other applications include nuclear medicine, basic research, environmental monitoring, and aerial surveys.
Sodium iodide can be used both as single crystals and as polycrystals for this purpose. The iodine provides most of the stopping power in sodium iodide (since it has a high Z = 53). These crystalline scintillators are characterized by high density, high atomic number, and pulse decay times of approximately 1 microsecond (~ 10-6 sec). The wavelength of maximum emission is 415 nm.
NaI(Tl) is the most widely used scintillation material.
Sodium iodide activated with a trace amount of thallium converts about 15% of fast particle energy into light, a figure that is the highest among commonly used scintillation materials. The high atomic number of the iodine component ensures that large crystals of sodium iodide will fully absorb the energy of a large fraction of all incident gamma rays. NaI(Tl) detectors have other advantages, very large crystals can be grown and raw material is not expensive, which makes high efficiency for gamma ray detection is easily achieved. Sodium Iodide Thallium-doped crystals have a very high luminescence (scintillation) efficiency and are available in a wide variety of sizes and geometries. Because of all its advatnages, NaI(Tl) detectors became very soon one of the most convenient options for many applications, from radiology to environmental monitoring.
Parameter
| Chemical formula | NaI |
| Molar mass | 149.894 |
| Appearance | white solid deliquescent |
| Crystal structure | Halite, cF8 |
| Space group | Fm3m, No. 225 |
| Lattice constant | a = 0.6462 nm |
| Formula units (Z) | 4 |
| Coordination geometry | Octahedral |
| Density (g/cm³) | 3.67 |
| Atomic Number (Effective) | 56 |
| Melting Point (ºK) | 924 |
| Boiling point (ºK) | 1557 |
| Thermal Expansion Coeff. (C⁻¹) | 47.4 x 10‾⁶ |
| Cleavage Plane | <100> |
| Hardness (Mho) | 2 |
| Hygroscopic | Yes |
| Solubility (g/100gH₂0) | TBA (23ºC) |
| Magnetic susceptibility (χ) | −57×10−6 cm3 mol−1 |
| Heat capacity (C) | 52.1 J mol−1 K−1 |
| Wavelength (Max. Emission) (nm) | 415 |
| Wavelength Range (nm) | 325 – 550 |
| Decay Time (ns) | 230 |
| Light Yield (photons/keV) | 55 |
| Photoelectron Yield (% of NaI) | 100 |
| Radiation Length (cm) | 2.6 |
| Optical Transmission (µm) | 0.15 – 12.5 |
| Transmittance (%) | >90 (0.35 – 9µm) |
| Refractive Index | 1.85 (@415nm) |
| Reflection Loss/Surface (%) | 6.8 |
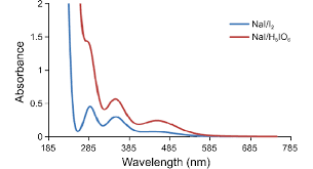 | 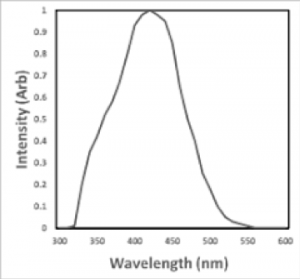 |
| NaI Absorption | NaI Intensity vs. wavelength |
- Are one of the ‘brightest’ scintillators available
- Have good density and a high Z
- Have an optical output well match to the maximum sensitivity of commonly available PMTs
- Have good ‘rad hard’ properties
- Do not glow in the UV
- Have an optical output largely independent of temperature
- Time-of-Flight measurements
- Positron lifetime studies
- Positron Emission Tomography (PET)
- Specialist applications in nuclear and high energy physics
- Used in high vibration environments
- Environmental monitoring
- aerial surveys
| [1] Chuong H D, Hung N Q , Le N , et al. Validation of gamma scanning method for optimizing NaI(Tl) detector model in Monte Carlo simulation[J]. Applied Radiation and Isotopes, 2019, 149:1-8. |
| [2] Ji Y Y ,Chung K H , Kang M J . Assessment of dose rate of detected gamma emitting nuclides using a carborne survey with a large volume NaI(Tl) detector[J]. Progress in Nuclear Energy, 123. |
| [3] Pablo, Mínguez, Emilia, et al. Analysis of activity uptake, effective half-life and time-integrated activity for low- and high-risk papillary thyroid cancer patients treated with 1.11GBq and 3.7GBq of 131I-NaI respectively – ScienceDirect[J]. Physica Medica, 2019, 65:143-149. |
| [4] Lugendo I J , Ahn J K , Kumwenda M J , et al. Lifetime measurement of the first excited 5 / 2 + state in 133Cs using NaI(Tl) and LaBr3(Ce) detectors[J]. Applied Radiation and Isotopes, 2019, 150:141-145. |
| [5] Almaz E , Akyol A . Stripping of the NaI (Tl) detector response function for continuous energy photon spectrum by SVD approach[J]. Nuclear Instruments and Methods in Physics Research Section B Beam Interactions with Materials and Atoms, 2020, 474:1-5. |
| [6] Li H B , Lei W G , Feng T C , et al. A NaI(Tl)-based radioactive noble-gas monitoring system used for radiation monitoring[J]. Applied Radiation and Isotopes, 2020, 163:109230. |
| [7] Jin W , Zhang Y , Liu D , et al. Automated spectra analysis of in situ radioactivity measurements in the marine environment using NaI(Tl) detector[J]. Applied Radiation and Isotopes, 2018, 141:S0969804318305013-. |
| [8] Pilakouta M , Pappa F K , Patiris D L , et al. A methodology for expanding the use of NaI(Tl) based spectrometry in environmental radioactivity measurements[J]. Applied Radiation and Isotopes, 2018, 139. |
| [9] Joo H W , Park H S , Kim J H , et al. Quenching factor measurement for NaI(Tl) scintillation crystal[J]. Astroparticle Physics, 2019, 108:50-56. |
| [10] Duc Tam H , Hai Yen N T , Tran L B , et al. Optimization of the Monte Carlo simulation model of NaI(Tl) detector by Geant4 code.[J]. Appl Radiat Isot, 2017:75-79. |
| [11] Zhang Y , Jiao Q , Ma B , et al. Structure and ionic conductivity of new Ga2S3-Sb2S3-NaI chalcogenide glass system[J]. Physica B Condensed Matter, 2019, 570(10). |
| [12] Syha C , Sm A , Hyla B , et al. Preliminary study on the detection efficiency and estimation of minimum detectable activity for a NaI(Tl)-based seawater monitoring system – ScienceDirect[J]. Journal of Environmental Radioactivity, 218. |
| [13] Arnquist I J , Hoppe E W . The quick and ultrasensitive determination of K in NaI using inductively coupled plasma mass spectrometry[J]. Nuclear Instruments & Methods in Physics Research A, 2017, 851(Apr.11):15-19. |
| [14] Kim J , Pa Rk K , Hwang J , et al. Efficient Design of a ∅2×2 Inch NaI(Tl) Scintillation Detector Coupled with a SiPM in an Aquatic Environment[J]. Nuclear Engineering and Technology, 2019. |
| [15] Singh P , Gupta P N , Saroj A L . Ion dynamics and dielectric relaxation behavior of PVA-PVP-NaI-SiO2 based nano-composites polymer blend electrolytes[J]. Physica B Condensed Matter, 2019, 578:411850. |
| [16] El-Gammal W , El-Kourghly K M , El-Tahawy M S , et al. A hybrid analytical-numerical method for full energy peak efficiency calibration of a NaI detector[J]. Nuclear Instruments and Methods in Physics Research Section A Accelerators Spectrometers Detectors and Associated Equipment, 2020:164181. |
| [17] Zebardastan N , Khanmirzaei M H , Ramesh S , et al. Presence of NaI in PEO/PVdF-HFP blend based gel polymer electrolytes for fabrication of dye-sensitized solar cells[J]. Materials Science in Semiconductor Processing, 2017, 66:144-148. |
| [18] X, Huyan, O, et al. Geant4 simulations of the absorption of photons in CsI and NaI produced by electrons with energies up to 4 MeV and their application to precision measurements of the β -energy spectrum with a calorimetric technique[J]. Nuclear Instruments and Methods in Physics Research, Section A. Accelerators, Spectrometers, Detectors and Associated Equipment, 2018, 879:134-140. |
| [19] Huang T , Fu Q , Lin S , et al. NaI(Tl) scintillator read out with SiPM array for gamma spectrometer[J]. Nuclear Instruments & Methods in Physics Research A, 2017, 851(Apr.11):118-124. |
| [20] Zou Q , Zheng S , Wang X , et al. Influenza A-associated severe pneumonia in hospitalized patients: Risk factors and NAI treatments[J]. International Journal of Infectious Diseases, 2020, 92. |
| [21] Han C A , SS A, Hx B , et al. Catalytic oxidative conversion of aldehydes into nitriles using NH3·H2O/FeCl2/NaI/Na2S2O8: A practical approach to febuxostat – ScienceDirect[J]. Tetrahedron Letters, 2019, 60( 21):1434-1436. |
| [22] Optimization of data acquisition parameters for single gamma line sensing: KSr_2I_5:Eu~(2+) and NaI(Tl) compared[J]. Nuclear Instruments & Methods in Physics Research, 2019, 938(SEP.11):36-40. |
| [23] Hendriks M , Erik V . 4f→5d and anomalous emission in Yb2+ doped NaI, SrI2 and LaI3 powders prepared by rapid melting and quenching in vacuum[J]. Journal of Luminescence, 2018, 207. |
| [24] Ramadhan R A , Abdullah M S . Background reduction by Cu/Pb shielding and efficiency study of NaI(TI) detector[J]. Nuclear Engineering & Technology, 2018:S1738573317306277. |
| [25] Mouhti I , Elanique A , Messous M Y , et al. Validation of a NaI(Tl) and LaBr 3 (Ce) detector’s models via measurements and Monte Carlo simulations[J]. Journal of Radiation Research & Applied ences, 2018:S1687850718300530-. |
| [26] Dinh C H , Le T , Vo N H , et al. A Revision Of The Virtual Point Detector Model For Calculating Nai(Tl) Detector Efficiency[J]. Applied Radiation and Isotopes, 2020, 162. |
| [27] Bartram C , Henning R , Primosch D . Demonstration of o-Ps detection with a cylindrical array of NaI detectors[J]. Nuclear Instruments & Methods in Physics Research. Section A, Accelerators, Spectrometers, Detectors and Associated Equipment, 2020, 966(Jun.21):163856.1-163856.10. |
| [28] X Li, Wang Z , Lv H , et al. Measurement of the energy of fast neutrons in the presence of gamma rays using a NaI(Tl) and a plastic scintillator[J]. Nuclear Instruments and Methods in Physics Research Section A Accelerators Spectrometers Detectors and Associated Equipment, 2020, 976:164257. |
| [29] Paradis H , Bobin C , J Bobin, et al. Spectral unmixing applied to fast identification of γ-emitting radionuclides using NaI(Tl) detectors[J]. Applied Radiation and Isotopes, 2020, 158(4):109068. |
| [30] Jyl A , Ga B , Ch C , et al. A Study of NaI(Tl) crystal encapsulation using organic scintillators for the dark matter search – ScienceDirect[J]. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment, 953. |





Leave a Reply